
Contains Nonbinding Recommendations
Food Labeling: Revision of the Nutrition and
Supplement Facts Labels: Guidance for
Industry
Small Entity Compliance Guide
Additional copies are available from:
Office of Nutrition and Food Labeling
Nutrition Programs Staff, HFS-830
Center for Food Safety and Applied Nutrition
Food and Drug Administration
5001 Campus Drive
College Park, MD 20740
(Tel) 240-402-1450
http://www.fda.gov/FoodGuidances
You may submit electronic or written comments regarding this guidance at any time. Submit
electronic comments to https://www.regulations.gov. Submit written comments to the Dockets
Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061,
Rockville, MD 20852. All comments should be identified with the docket number listed in the
notice of availability that publishes in the Federal Register.
For questions regarding this draft document contact the Center for Food Safety and Applied
Nutrition (CFSAN) at 240-402-1450.
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Food Safety and Applied Nutrition
January 2020
Contains Nonbinding Recommendations
Table of Contents
I. Introduction
II. Who Is Subject to the Rule?
III. What Foods Are Covered by the Rule?
III.A Does the Rule Cover Foods for the General Food Supply?
III.B Does the Rule Cover Foods for Infants and Young Children 1 Through 3 Years
of Age?
III.C Does the Rule Cover Foods for Pregnant Women and Lactating Women?
III.D Does the Rule Cover Dietary Supplements?
IV. What Foods Are Not Covered by the Rule?
V. Which Nutrients Must Newly be Declared, and What Changes
Have Been Made to Nutrients Previously Required or Allowed to
be Declared?
V.A Which Nutrients Are Newly Required to be Declared?
V.B Have There Been Any Changes to the Definition or Presentation of Nutrients
That Were Already Required to Be Declared?
V.C Which Nutrients Can I Still Voluntarily Declare, Even Though They Are No
Longer Mandatory?
V.D Which Nutrients Are Newly Allowed to be Voluntarily Declared?
V.E Which Nutrients Can I No Longer Declare?
VI. How Do I Comply with the Recordkeeping Requirements?
VI.A When Are Records Necessary?
VI.B What Counts as a Record?
VI.C How Long Must Records be Kept?
Contains Nonbinding Recommendations
VII. How Have the Values of Nutrients Been Updated?
VII.A How Have the Calculations of Caloric Content Changed?
VII.B How Have the Daily Reference Values (DRVs) Changed?
VII.C How Have the Reference Daily Intakes (RDIs) Changed?
VII.D How Do the Nutrient Requirements for Supplement Facts Labels Differ?
VII.E Has Nutrient Compliance or the Level of Variance Allowed Changed?
VII.F Which Version of the Official Methods of Analysis of the AOAC International
Should be Used?
VIII. How Do I Comply with the Formatting Requirements?
VIII.A How Do I Comply with the Standard Version of the Nutrition Facts Label?
VIII.B When Can I Use the Tabular Format?
VIII.C How Do I Comply with These Requirements if My Food Packaging Only Has a
Total Surface Area Available to Bear Labeling of 40 or Less Square Inches?
VIII.D How Do I Comply with the Nutrition Labeling Formatting Requirements when
Dual Column Labeling is Required or Provided Voluntarily?
VIII.E When Can the Simplified Format Be Used?
VIII.F Are Foods in Small Packages with a Total Surface Area Available to Bear
Labeling of Less than 12 Square Inches Exempt from the Nutrition Facts
Labeling Requirements?
VIII.G How Do These New Formatting Rules Affect the Percentage Juice Declaration?
VIII.H Are There Specific Formatting Requirements for Supplement Facts Labels?
IX. When Must I Comply with the Rule?
X. Why Must I Comply with the Rule?
XI. References

Contains Nonbinding Recommendations
1
Food Labeling: Revision of the Nutrition and
Supplement Facts Labels: Guidance for
Industry
1
Small Entity Compliance Guide
This guidance represents the current thinking of the Food and Drug Administration (FDA or we)
on this topic. It does not establish any rights for any person and is not binding on FDA or the
public. You can use an alternative approach if it satisfies the requirements of the applicable
statutes and regulations. To discuss an alternative approach, contact the FDA staff responsible
for this guidance as listed on the title page.
I. Introduction
In the Federal Register of May 27, 2016 (81 FR 33742), FDA (we) published a final rule entitled
“Food Labeling: Revision of the Nutrition and Supplement Facts Labels” (“the final rule”). The
final rule amends the labeling regulations for conventional foods and dietary supplements to
provide updated nutrition information on the label to assist consumers in maintaining healthy
dietary practices and set a compliance date of July 26, 2018, for manufacturers with $10 million
or more in annual food sales, and July 26, 2019, for manufacturers with less than $10 million in
annual food sales. We subsequently extended the compliance dates to January 1, 2020, and
January 1, 2021, respectively (83 FR 19619). We have prepared this Small Entity Compliance
Guide in accordance with section 212 of the Small Business Regulatory Enforcement Fairness
Act (Public Law 104-121, as amended by Public Law 110-28). This guidance document restates
in plain language the revisions made in the final rule and is intended to help small entities
comply with the requirements established in 21 CFR 101.9, 101.30, and 101.36.
FDA’s guidance documents, including this guidance, do not establish legally enforceable
responsibilities. Instead, guidances describe our current thinking on a topic and should be
viewed only as recommendations, unless specific regulatory or statutory requirements are cited.
The use of the word should in our guidances means that something is suggested or
recommended, but not required.
In the remainder of this guidance, “you” and “I” refer to food manufacturers that are subject to
the rule. Many answers in this guidance are followed by citations to show where a specific
requirement can be found in either the Federal Food, Drug, and Cosmetic Act (FD&C Act) or
Title 21 of the Code of Federal Regulations.
1
This guidance has been prepared by the Office of Nutrition and Food Labeling in the Center for Food Safety and
Applied Nutrition at the U.S. Food and Drug Administration.
Contains Nonbinding Recommendations
2
II. Who Is Subject to the Rule?
You are subject to the rule if you manufacture food that is subject to our nutrition labeling
requirements. The nutrition labeling requirements apply to both conventional foods under 21
CFR 101.9(a) and dietary supplements under 21 CFR 101.9(j)(6).
III. What Foods Are Covered by the Rule?
III.A Does the Rule Cover Foods for the General Food Supply?
Yes. Foods for the general food supply are foods eaten by persons 4 years of age and older.
III.B Does the Rule Cover Foods for Infants and Young Children 1 Through 3 Years of
Age?
Yes. Foods, other than infant formula, represented or purported to be specifically for infants
through 12 months of age and children 1 through 3 years of age are subject to nutrition labeling
(21 CFR 101.9(j)(5)(i)). Manufacturers of foods represented or purported to be specifically for
infants through 12 months and/or children 1 through 3 years of age must use the Reference Daily
Intakes (RDIs) and Daily Reference Values (DRVs) that are specified for this intended group
when calculating percent Daily Values (DVs) for labels (21 CFR 101.9(c)(8) and (9)). The
previous categories of “infants” (or “infants 7 to 12 months”) and “children less than 4 years”
have been changed to “infants through 12 months” and “children 1 through 3 years of age”
throughout 21 CFR 101.9.
III.C Does the Rule Cover Foods for Pregnant Women and Lactating Women?
Yes. Manufacturers of foods represented or purported to be specifically for pregnant women and
lactating women must use the RDIs and DRVs that are specified for this intended group when
calculating percent DVs for labels (21 CFR 101.9(c)(8) and (9)).
III.D Does the Rule Cover Dietary Supplements?
Yes. Section 201(f) of the FD&C Act defines “food” as: “(1) articles used for food or drink for
man or other animals, (2) chewing gum, and (3) articles used for components of any such
article.” Further, section 201(ff) of the FD&C Act explains that dietary supplements are deemed
to be foods within the meaning of the FD&C Act except for the purposes of sections 201(g)
(definition of “drug”) and 417 (reportable food registry) of the FD&C Act. Nutrition labeling
information for food must be provided for all products intended for human consumption and
offered for sale, unless an exemption is provided (21 CFR 101.9(a)). As dietary supplements fall
under the definition of “food,” they are therefore subject to nutrition labeling. Specific nutrition
labeling requirements and guidelines for dietary supplements can be found in 21 CFR 101.36.
IV. What Foods Are Not Covered by the Rule?
Contains Nonbinding Recommendations
3
Under 21 CFR 101.9(j), numerous foods are exempt from nutrition labeling requirements or are
subject to special labeling requirements. Such products generally include: (1) foods offered for
sale by a retailer who has annual gross sales made or business done in sales to consumers that is
not more than $500,000; (2) foods offered for sale by a retailer who has annual gross sales made
or business done in sales of food to consumers of not more than $50,000; (3) medical foods; and
(4) foods that contain insignificant amounts of all nutrients (e.g., coffee beans, tea leaves). For
more information on foods not covered by the rule and for further information about exemptions
from these requirements, see 21 CFR 101.9(j).
V. Which Nutrients Must Newly be Declared, and What Changes
Have Been Made to Nutrients Previously Required or Allowed to be
Declared?
V.A Which Nutrients Are Newly Required to be Declared?
V.A.1 Added Sugars
V.A.1.(a) How Are Added Sugars Defined?
Added sugars are defined as sugars that are “either added during the processing of foods, or are
packaged as such, and include sugars (free, mono- and disaccharides), sugars from syrups and
honey, and sugars from concentrated fruit or vegetable juices that are in excess of what would be
expected from the same volume of 100 percent fruit or vegetable juice of the same type” (21
CFR 101.9(c)(6)(iii)). This definition includes single ingredient foods, such as individually
packaged table sugar (see Section V.A.1.(a).(i) and Ref. 1).
The following do not fall under the definition of added sugars:
• Sugars in fruit or vegetable juice concentrated from 100 percent juices that are sold to
consumers (e.g., frozen 100 percent fruit juice concentrate) (21 CFR 101.9(c)(6)(iii));
• Sugars in fruit juice concentrates that are used to formulate the fruit component of
jellies, jams, or preserves in accordance with the standards of identities set forth in 21
CFR 150.140 and 150.160 (21 CFR 101.9(c)(6)(iii));
• Sugars in the fruit component of fruit spreads (21 CFR 101.9(c)(6)(iii));
• Sugar alcohols;
• Sugars in juice concentrates that are counted towards percentage juice label declaration
under 21 CFR 101.30 for 100 percent juice or 21 CFR 102.33 for juice beverages (21
CFR 101.9(c)(6)(iii));
• Sugars in juice concentrates that are used to standardize the Brix values of a single
species juice consisting of juice directly expressed from a fruit or vegetable in
accordance with 21 CFR 102.33(g)(2) (21 CFR 101.9(c)(6)(iii));
• Naturally-occurring sugars found in milk and dairy ingredients, except lactose as defined
in 21 CFR 168.122; and
Contains Nonbinding Recommendations
4
• Naturally-occurring sugars found in whole fruits and vegetables or dried fruits which
have not had any sugar added to them.
The amount of added sugars declared on the label should never exceed the amount of total sugars
on the label.
V.A.1.(a).(i) Do Honey, Maple Syrup, and Other Single-Ingredient Sugars and Syrups Have to
be Declared as Added Sugars?
In the Federal Register of June 20, 2019 (84 FR 28726), we announced the availability of a final
guidance for industry entitled “The Declaration of Added Sugars on Honey, Maple Syrup, Other
Single-Ingredient Sugars and Syrups, and Certain Cranberry Products,” which provides clarity
on the labeling of added sugars for such products (Ref. 1). In that guidance, we discuss section
12516 of the Agriculture Improvement Act of 2018 (Pub. L. 115-334) (“the Farm Bill”), which
states that the food labeling requirements cannot require the declaration “Includes Xg Added
Sugars” in a serving of these single-ingredient products. While these products are not required to
have this declaration of the gram amount of added sugars, the Farm Bill did not change the
requirement under the Nutrition Facts label final rule to include the percent DV for the
contribution of sugars from these products to the added sugars in the diet, so the percent DV for
added sugars must still be included consistent with 21 CFR 101.9(d)(7)(ii). We stated that we
intend to exercise enforcement discretion by permitting the use of the “†” symbol immediately
following the percent DV declaration for added sugars on packages and containers of single-
ingredient sugars and syrups. This symbol would lead to a footnote inside the Nutrition Facts
label, explaining the amount of added sugars that one serving of the product contributes to the
diet, as well as the contribution of a serving of the product toward the percent DV for added
sugars. This symbol and footnote are not required; however, we encourage you to use them.
V.A.1.(a).(ii) Do Added Sugars in Dried Cranberry Products and Cranberry Beverage Products
Have to be Declared as Added Sugars?
You must still declare added sugars in grams and the corresponding percent DV on labels for all
dried cranberry products and cranberry beverage products (21 CFR 101.9(c)(6)(iii)). In the
Declaration of Added Sugars on Honey, Maple Syrup, Other Single-Ingredient Sugars and
Syrups, and Certain Cranberry Products guidance, we said that we intend to exercise
enforcement discretion for these cranberry products to allow the use of a symbol immediately
following the added sugars percent DV declaration which would lead to a truthful and not
misleading statement outside the Nutrition Facts label explaining that sugars are added to
improve the palatability of naturally tart cranberries (see Ref. 1). This symbol can be used for
cranberry products that are sweetened with added sugars and that contain total sugars per serving
at levels no greater than comparable products with no added sugars (i.e. unsweetened grape
juice) (Ref. 1).
V.A.1.(a).(iii) Does Allulose Count as an Added Sugar?
The final rule does not reach a decision as to whether allulose should be excluded from the
labeling of carbohydrate, sugars, and/or added sugars. We stated that, as a monosaccharide, it
Contains Nonbinding Recommendations
5
must be included in the declaration of each, pending any future rulemaking that would otherwise
exclude it from the declaration (81 FR 33742 at 33796; 21 CFR 101.9(c)(i) through (iii)).
In the Federal Register of April 18, 2019 (84 FR 16272), we announced the availability of a
draft guidance for industry entitled “The Declaration of Allulose and Calories from Allulose on
Nutrition and Supplement Facts Labels.” This guidance, when finalized, would advise
manufacturers of our intent to exercise enforcement discretion for the exclusion of allulose from
the amount of “Total Sugars” and “Added Sugars” declared on the label and the use of a general
factor of 0.4 calories per gram for allulose when determining “Calories” on the Nutrition and
Supplement Facts labels, pending review of the issues in a rulemaking (Ref. 2).
V.A.1.(b) How Do I Calculate Added Sugars?
In the Federal Register of November 5, 2018 (83 FR 55266), we announced the availability of a
final guidance for industry entitled “Nutrition and Supplement Facts Labels: Questions and
Answers Related to the Compliance Date, Added Sugars, and Declaration of Quantitative
Amounts of Vitamins and Minerals.” This guidance provides questions and answers (Q&A) on
topics related to compliance with the labeling of added sugars. The Q&A discusses how you
should determine the amount of added sugars in a serving of a product, as well as which
ingredients need to be taken into consideration when calculating the added sugars declaration for
a serving of a product (Ref. 3).
This guidance also provides a detailed discussion on the calculation of added sugars for
ingredients and products such as concentrated fruit and vegetable purees, fruit and vegetable
pastes, fruit and vegetable powders, juice cocktails, or juice blends, which often contain juice
concentrates (Q&A IV. 7-12), products for which manufacturers employ a hydrolysis step (Q&A
IV. 14-16), and products that undergo non-enzymatic browning or fermentation (Q&A IV. 18–
21) (Ref. 3). Please refer to this guidance for questions you may have regarding the calculation
and declaration of added sugars for a serving of your product.
V.A.2 Vitamin D
Vitamin D is now considered a nutrient of “public health significance” for the general
population, and its declaration is now mandatory (81 FR 33742 at 33884). Both the gram
amount and the percent DV for Vitamin D must be declared at the bottom of the label, directly
preceding the calcium declaration (21 CFR 101.9(c)(8)(ii)). While only the term “vitamin D”
can be used on the food labels (21 CFR 101.9(c)(8)(iv)), the specific form that is added to a food
must be listed in the ingredient list statement (21 CFR 101.4). For Supplement Facts labels, the
source ingredient may be identified within the nutrition label in parentheses immediately
following or indented beneath the name of a dietary ingredient and preceded by the word “as” or
“from” (21 CFR 101.36(d)). When you do not identify a source ingredient within the nutrition
label, you must list it in an ingredient statement (21 CFR 101.4(g)). You should not list it in both
places (81 FR 33742 at 33891).
Contains Nonbinding Recommendations
6
V.A.3 Potassium
Potassium is also now considered a nutrient of “public health significance” for the general
population (81 FR 33742 at 33884). It has been assigned an RDI, instead of a DRV (21 CFR
101.9(8)(iv)). We now require the declaration of both the gram amount and the percent DV for
potassium at the bottom of the label, directly following the iron declaration. Potassium is now
covered under the term “mineral” that appears in each section of 21 CFR 101.9. Any listing of
potassium on the Nutrition Facts label must meet the specific nutrient declaration requirements
for minerals under 21 CFR 101.9(g)(4), 101.9(g)(4)(i), 101.9(g)(4)(ii), and 101.9(g)(6). These
requirements are discussed further in section VII.E “Has Nutrient Compliance or the Level of
Variance Allowed Changed?” below.
V.B Have There Been Any Changes to the Definition or Presentation of Nutrients That
Were Already Required to Be Declared?
V.B.1 Dietary Fiber
Any dietary fiber declared on the label must meet the new definition of dietary fiber (21 CFR
101.9(c) and (c)(6)(i)), which is discussed in the following section. This definition includes a
listing of dietary fiber that FDA has determined should be in the calculation of dietary fiber for
declaration on Nutrition Facts labels. The new definition and process to amend the listing of
dietary fiber is discussed in this section.
V.B.1.(a) How Is Dietary Fiber Defined?
Dietary fiber is now defined as “non-digestible soluble and insoluble carbohydrates (with 3 or
more monomeric units), and lignin that are intrinsic and intact in plants; isolated or synthetic
non-digestible carbohydrates (with 3 or more monomeric units) determined by FDA to have
physiological effects that are beneficial to human health” (21 CFR 101.9(c)(6)(i)). Soluble fiber
and insoluble fiber both must meet this new definition of dietary fiber (21 CFR 101.9(c)(6)(i)(A)
and (B)).
V.B.1.(a).(i) Which Isolated or Synthetic Non-Digestible Carbohydrate(s) Qualify as Dietary
Fiber?
The following isolated or synthetic non-digestible carbohydrate(s) should be included in the
calculation of the amount of dietary fiber, as FDA has determined that they have physiological
effects that are beneficial to human health:
• Beta-glucan soluble fiber (as described in 21 CFR 101.81(c)(2)(ii)(A));
• Psyllium husk (as described in 21 CFR 101.81(c)(2)(ii)(A)(6));
• Cellulose;
• Guar gum;
• Pectin;
• Locust bean gum; and
• Hydroxypropylmethylcellulose. (21 CFR 101.9(c)(6)(i)).

Contains Nonbinding Recommendations
7
In the Federal Register of June 15, 2018 (83 FR 27894), we announced the availability of a final
guidance for industry entitled “The Declaration of Certain Isolated or Synthetic Non-Digestible
Carbohydrates as Dietary Fiber on Nutrition and Supplement Facts Labels,” where we stated that
we intend to propose that the following eight non-digestible carbohydrates be added to the
definition of dietary fiber, as well:
• Mixed plant cell wall fibers (a broad category that includes fibers like sugar cane fiber
and apple fiber, among many others);
• Arabinoxylan;
• Alginate;
• Inulin and inulin-type fructans;
• High amylose starch (resistant starch 2);
• Galactooligosaccharide;
• Polydextrose; and
• Resistant maltodextrin/dextrin (see Ref. 4).
Until we complete rulemaking to add any additional non-digestible carbohydrates to the
regulatory definition of dietary fiber, we intend to exercise enforcement discretion to allow
manufacturers to include the eight recognized fibers when calculating the amount of dietary fiber
to declare on the Nutrition and Supplement Facts labels.
Additionally, in March of 2019 and January of 2020, we granted citizen petition requests and
announced our intent to propose that “cross-linked phosphorylated RS4” and glucomannan,
respectively, be added to the definition of dietary fiber (Refs. 5 and 6). For up-to-date
information on the additional non-digestible carbohydrates that FDA intends to propose to be
added to the definition of dietary fiber, see “Questions and Answers on Dietary Fiber,” available
at http://www.fda.gov/food/food-labeling-nutrition/questions-and-answers-dietary-
fiber#synthetic_fibers.
V.B.1.(a).(ii) What If There is an Additional Isolated or Synthetic Non-Digestible Carbohydrate
I Want to be Included in the List of Dietary Fibers?
There are two ways that the list of dietary fiber can be amended to include additional isolated or
synthetic non-digestible carbohydrates in the definition. The first way is by submitting a citizen
petition to request an amendment to the dietary fiber definition (see 21 CFR 10.30(b)). The
second way involves the petition process for the authorization of a health claim. For further
information regarding the petition process for the authorization of a health claim, refer to 21 CFR
101.70. Once an isolated or synthetic non-digestible carbohydrate is added to the dietary fiber
definition, all manufacturers must include it as part of the total dietary fiber declaration if it is
present in their product (21 CFR 101.9(c)(6)(i)).
In the Federal Register of March 2, 2018 (83 FR 8997), we announced the availability of a final
guidance entitled “Scientific Evaluation of the Evidence on the Beneficial Physiological Effects
of Isolated or Synthetic Non-Digestible Carbohydrates Submitted as a Citizen Petition (21 CFR
10.30)” (Ref. 7). For further information on this topic, please refer to that final guidance, as it
Contains Nonbinding Recommendations
8
goes further in explaining FDA’s current thinking on information needed when submitting a
citizen petition and the scientific review approach we plan to use for evaluating scientific
evidence to determine whether an isolated or synthetic non-digestible carbohydrate that is added
to food has a physiological effect that is beneficial to human health.
V.B.1.(b) How Do I Calculate Dietary Fiber?
Compliance with any declaration of dietary fiber is determined using the appropriate “Official
Methods of Analysis of the AOAC International” (21 CFR 101.9(g)(2)). AOAC 2009.01,
AOAC 2011.25, or an equivalent AOAC method of analysis may be used to measure the amount
of dietary fiber in a serving of a product. The methods used must support the dietary fiber
definition and therefore must measure lower molecular weight non-digestible oligosaccharides
(DP 3-9) if present in a food (see 21 CFR 101.9(c)(6)(i)). We consider AOAC 2009.01 and
AOAC 2011.25 to be reliable and appropriate methods to measure the amount of dietary fiber in
a serving of a product. AOAC 2011.25 is a newer method that can measure low molecular
weight non-digestible carbohydrates and is considered a reliable and appropriate method to
measure the amount of soluble and insoluble fiber in a serving of a product, if declared
separately (81 FR 33742 at 33960).
If a food contains a mixture of non-digestible carbohydrates that do and do not meet the
proposed dietary fiber definition, and the label of the food declares dietary fiber content, then
you must make and keep records to verify the amount of non-digestible carbohydrates that do not
meet the proposed definition of dietary fiber that have been added to the food (21 CFR
101.9(g)(10)(i) through (iii); see also section VI. “How Do I Comply with the Recordkeeping
Requirements?”).
V.B.2 Total Sugars
“Sugars” has changed to “Total Sugars,” which includes both “Added Sugars” and sugars that
are naturally occurring in food. The label declaration of “Total Sugars” is not required if the
product contains less than one gram of sugars per serving and no claims are made about sugars,
sweeteners, or sugar alcohol content. If the total sugars content is not required and therefore not
declared, the statement “Not a significant source of total sugars” must be placed at the bottom of
the table of nutrient values (see 21 CFR 101.9(c)(6)(ii)).
V.B.3 Sugar Alcohol
In addition to preexisting regulations, sugar alcohol content must now also be declared when a
claim is made on the label or in labeling about added sugars when sugar alcohols are present in
the food (21 CFR 101.9(c)(6)(iv)).
V.C Which Nutrients Can I Still Voluntarily Declare, Even Though They Are No Longer
Mandatory?
While the declarations of vitamins A and C are no longer mandatory, you can declare this
information voluntarily. Declaration of vitamins A and C can be provided for foods for adults
Contains Nonbinding Recommendations
9
and children 4 years or more of age, as well as on foods represented or purported to be
specifically for infants through 12 months, children 1 through 3 years of age, pregnant women,
and lactating women. However, vitamin A and vitamin C declaration is mandatory when the
respective nutrient is added as a nutrient supplement or claims are made about it on the label or
in the labeling of foods (21 CFR 101.9(c)(8)(ii)). There have been no changes to the prior
provision that allowed for voluntary declaration of the percent of vitamin A that is present as β-
carotene (101.9(c)(8)(vi)).
V.D Which Nutrients Are Newly Allowed to be Voluntarily Declared?
Declaration of fluoride is voluntary whether it is intentionally added or naturally present, unless
a claim is made about fluoride content, which would trigger mandatory declaration. When
fluoride content is declared, it must be expressed as zero when a serving contains less than 0.1mg
of fluoride, to the nearest 0.1mg increment when a serving contains less than or equal to 0.8mg
of fluoride, and the nearest 0.2mg when a serving contains more than 0.8mg of fluoride (21 CFR
101.9(c)(5)).
For bottled water that bears a statement about added fluoride, as permitted under 21 CFR
101.13(q)(8), nutrition labeling that complies with the simplified format requirements (21 CFR
101.9(f)) is required (21 CFR 101.9(c)(5)).
V.E Which Nutrients Can I No Longer Declare?
V.E.1 “Calories From Fat”
While the declaration of “Calories from fat” used to be required, it is no longer required nor is it
allowed to be declared voluntarily (81 FR 33742 at 33780). Any reference to this declaration has
been removed from the updated regulations (81 FR 33742 at 33780).
V.E.2 “Other Carbohydrate”
The voluntary declaration of “Other Carbohydrate” is also no longer allowed. Any reference to
this declaration has been removed from the updated regulations (81 FR 33742 at 33868).
VI. How Do I Comply with the Recordkeeping Requirements?
VI.A When Are Records Necessary?
For certain nutrients, there are no AOAC official methods of analysis or other reliable or
appropriate analytical procedures available to verify the amount of the declared nutrient on the
Nutrition Facts or Supplement Facts label. Verification is important because it ensures that the
declared nutrient amount is truthful, accurate, and complies with all applicable labeling
requirements. In such situations, we require you to make and keep records that are necessary to
verify the declaration of these nutrients, as you are in the best position to verify how you arrived
at this determination and to know which of your records provide the necessary documentation for
us to determine compliance.
Contains Nonbinding Recommendations
10
Records are needed to verify the declaration of the following:
• The amount of added sugars added to the food during processing, when both naturally
occurring and added sugars are present in a food (21 CFR 101.9(g)(10)(iv));
• The amount of added sugars, if packaged as a separate ingredient, as packaged (whether
as part of a package containing one or more ingredients or packaged as a single
ingredient) when both naturally occurring and added sugars are present in a food (21
CFR 101.9(g)(10)(iv));
• The amount of added non-digestible carbohydrate(s) that does not meet the definition of
dietary fiber when the dietary fiber present in a food is a mixture of non-digestible
carbohydrates that do and that do not meet the definition of dietary fiber (21 CFR
101.9(g)(10)(i));
• The amount of added soluble non-digestible carbohydrate(s) that does not meet the
definition of dietary fiber when the soluble dietary fiber present in a food is mixture of
soluble non-digestible carbohydrates that do and that do not meet the definition of
dietary fiber (21 CFR 101.9(g)(10)(ii));
• The amount of added insoluble non-digestible carbohydrate(s) that does not meet the
definition of dietary fiber when the insoluble dietary fiber present in a food is a mixture
of insoluble non-digestible carbohydrates that do and that do not meet the definition of
dietary fiber (21 CFR 101.9(g)(10)(iii));
• The amount of all rac-α-tocopherol added to the food and RRR-α-tocopherol in the
finished food when a mixture of both forms of vitamin E are present in a food (21 CFR
101.9(g)(10)(vi)); and
• The amount of synthetic folate and/or folic acid added to the food and the amount of
naturally-occurring folate in the finished food when a mixture of both forms is present in
a food (21 CFR 101.9(g)(10)(vii)).
For example, if you manufacture products containing fruit and vegetable juice concentrates as an
ingredient that have not been reconstituted to 100 percent juice in the finished food, you must
provide documentation that shows how you determined how much of the sugars provided by the
juice concentrate should be declared as added sugars in the finished product (21 CFR
101.9(g)(10)(iv)).
Similar verification is needed regarding the amount of added sugars in specific foods, alone or in
combination with naturally occurring sugars, where the added sugars are subject to non-
enzymatic browning and/or fermentation. In such situations, you must:
1. make and keep records of all relevant scientific data and information relied upon to
demonstrate the amount of added sugars in the food after non-enzymatic browning and/or
fermentation, and a narrative explaining why the data and information are sufficient to
demonstrate the amount of added sugars declared in the finished food, provided the data
and information used is specific to the type of food manufactured; or
2. make and keep records of the amount of sugars added to the food before and during the
processing of the food, and if packaged as a separate ingredient, as packaged (whether as
part of a package containing one or more ingredients or packaged as a single ingredient)
Contains Nonbinding Recommendations
11
and in no event will the amount of added sugars declared exceed the amount of total
sugars on the label; or
3. submit a citizen petition requesting alternative means of compliance. The petition must
provide scientific data or other information for why the amount of added sugars in a
serving of the product is likely to have a significant reduction in added sugars compared
to the amount added prior to non-enzymatic browning and/or fermentation. A significant
reduction is a reduction in added sugars after non-enzymatic browning and/or
fermentation that may be significant enough to impact the label declaration for added
sugars by an amount that exceeds the reasonable deficiency acceptable within good
manufacturing practice under 101.9(g)(6). You must include the reason that you are
unable to determine a reasonable approximation of the amount of added sugars in a
serving of your finished product and a description of the process that you used to come to
that conclusion.
(See 21 CFR 101.9(g)(10)(v)(A) through (C).)
VI.B What Counts as a Record?
Records can include, for example, analyses of databases, recipes or formulations, or batch
records (21 CFR 101.9(g)(10)). They can be kept either as original records, true copies (e.g.
photocopies, pictures, scanned copies), or electronic records in accordance with 21 CFR part 11
(21 CFR 101.9(g)(11)). The records requirements provide flexibility in what records you make
available to FDA to verify the declared amounts of these nutrients. As our regulations pertaining
to disclosure of public information, at part 20, include provisions that protect trade secrets and
commercial or financial information which is privileged or confidential, you should mark the
information as such before providing the records to FDA (81 FR 33742 at 33962).
VI.C How Long Must Records be Kept?
Records must be kept for a period of two years after introduction or delivery for introduction of
the food into interstate commerce, and they must be provided to FDA upon request during an
inspection for official review and copying (or other means of reproduction) (21 CFR
101.9(g)(11)). Records need to be reasonably accessible to FDA during an inspection at each
manufacturing facility, even if not stored onsite, to determine compliance with labeling
requirements (81 FR 33742 at 33963). These recordkeeping requirements are the same for
foreign and domestic firms.
VII. How Have the Values of Nutrients Been Updated?
VII.A How Have the Calculations of Caloric Content Changed?
While the final rule still allows for the calculation of caloric content using a variety of methods,
there has been an update to the prior method of using the general factors of 4, 4, and 9 calories
per gram for protein, total carbohydrate less the amount of insoluble dietary fiber, and total fat,
respectively. Now, as stated in 21 CFR 101.9(c)(1)(i)(C), calories from carbohydrate are
required to be calculated using a general factor of 4 kcal/gram of total carbohydrate less the

Contains Nonbinding Recommendations
12
amount of non-digestible carbohydrates, which includes soluble and insoluble non-digestible
carbohydrates that do and do not meet the definition of dietary fiber, and sugar alcohols. A
general factor of 2 calories per gram is to be used for soluble non-digestible carbohydrates, but
insoluble non-digestible carbohydrates (0 calories per gram) are not included in the caloric
calculation. The calorie contribution of soluble non-digestible carbohydrate would be added to
that sum to determine the total carbohydrate calorie contribution (81 FR 33742 at 33867). The
following general factors are to be used for the caloric value of sugar alcohols:
• Isomalt – 2.0 calories per gram;
• Lactitol – 2.0 calories per gram;
• Xylitol – 2.4 calories per gram;
• Maltitol – 2.1 calories per gram;
• Sorbitol – 2.6 calories per gram;
• Hydrogenated starch hydrolysates – 3.0 calories per gram;
• Mannitol – 1.6 calories per gram; and
• Erythritol – 0 calories per gram (21 CR 101.9(c)(1)(i)(F)).
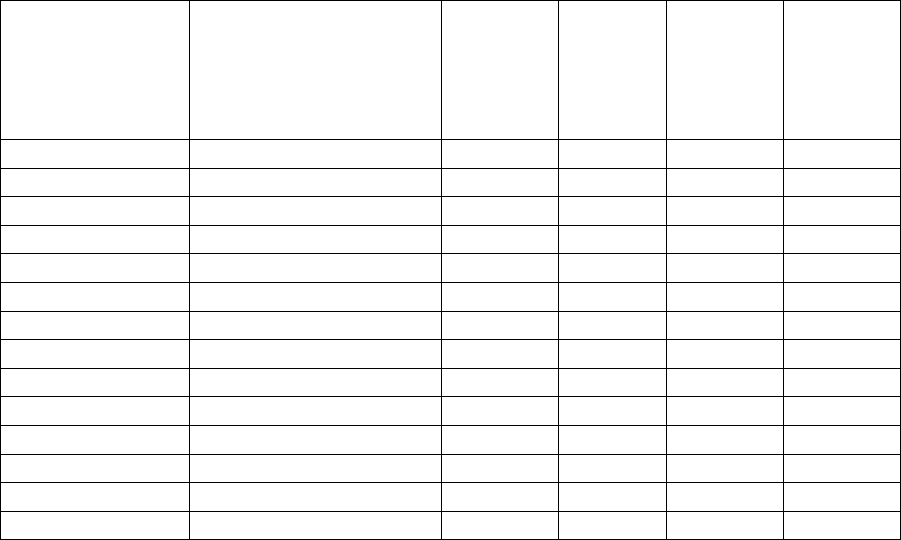
VII.B How Have the Daily Reference Values (DRVs) Changed?
The following table (Figure 1) reflects the updated DRVs for each population group that have
been established for fat, saturated fat, cholesterol, sodium, total carbohydrate, dietary fiber,
protein, and added sugars (see also 21 CFR 101.9(c)(9)).
Figure 1: Table 1 – Daily Reference Values Used to Calculate Percent DV
Nutrient
Unit of Measure
Adults
and
Children
≥ 4 years
Infants
through
12
months
Children
1
through
3 years
Pregnant
women and
lactating
women
Fat
Grams (g)
78
1
30
39
2
78
1
Saturated Fat
Grams (g)
20
1
N/A
10
2
20
1
Cholesterol
Milligrams (mg)
300
N/A
300
300
Sodium
Milligrams (mg)
2,300
N/A
1,500
2,300
Total
Carbohydrate
Grams (g)
275
1
95
150
2
275
1
Dietary Fiber
Grams (g)
28
1
N/A
14
2
28
1
Protein
Grams (g)
50
1
N/A
13
2
N/A
Added Sugars
Grams (g)
50
1
N/A
25
2
50
1
1
Based on the reference caloric intake of 2,000 calories for adults and children aged 4 years and older, and for
pregnant women and lactating women.
2
Based on the reference caloric intake of 1,000 calories for children 1 through 3 years of age.
We did not establish a reference calorie intake for infants through 12 months of age. We also did
not establish DRVs for trans fat, polyunsaturated fat, monounsaturated fat, insoluble fiber, total
sugars, or sugar alcohol for any population group.

Contains Nonbinding Recommendations
13
VII.C How Have the Reference Daily Intakes (RDIs) Changed?
Certain RDIs used in the declaration of the percent DV of nutrients on Nutrition and Supplement
Facts labels have been updated, which may affect the percent DV declared on such labels.
2
Figure 2 below reflects the list of ordered nutrients and their RDIs, which are used to calculate
the percent DVs (see also 21 CFR 101.9(c)(8)(iv)). RDIs have now been established for infants
through 12 months, children 1 through 3 years of age, and pregnant women and lactating women
for the following nutrients: vitamin A, vitamin D, vitamin E, vitamin C, vitamin K, vitamin B12,
vitamin B6, folate, choline, riboflavin, niacin, calcium, iron, thiamin, biotin, pantothenic acid,
phosphorus, iodine, magnesium, zinc, selenium, copper, manganese, chromium, molybdenum,
chloride, potassium (21 CFR 101.9(c)(8)(i) through (iv)). While we have not established a DRV
for protein for infants through 12 months or for pregnant women and lactating women, we have
established an RDI for protein of 11 grams and 71 grams, respectively, for these groups (see
Figure 2). We did not establish DVs for infants less than 7 months of age. Therefore, nutrition
information on foods purported for infants less than 7 months would not reflect DVs for that age
group (81 FR 33742 at 33917).
Figure 2: Table 2 – Reference Daily Intakes Used to Calculate Percent DV
Nutrient
Unit of Measure
Adults
and
Children
≥ 4 years
Infants
1
through
12
months
Children
1
through
3 years
Pregnant
women
and
lactating
women
Vitamin D
Micrograms (mcg)
20
10
15
15
Calcium
Milligrams (mg)
1,300
260
700
1,300
Iron
Milligrams (mg)
18
11
7
27
Potassium
Milligrams (mg)
4,700
700
3,000
5,100
Vitamin A
Micrograms RAE
2
(mcg)
900
500
300
1,300
Vitamin C
Milligrams (mg)
90
50
15
120
Vitamin E
Milligrams (mg)
3
15
5
6
19
Vitamin K
Micrograms (mcg)
120
2.5
30
90
Thiamin
Milligrams (mg)
1.2
0.3
0.5
1.4
Riboflavin
Milligrams (mg)
1.3
0.4
0.5
1.6
Niacin
Milligrams NE
4
(mg)
16
4
6
18
Vitamin B
6
Milligrams (mg)
1.7
0.3
0.5
2
Folate
5
Micrograms DFE
6
(mcg)
400
80
150
600
Vitamin B
12
Micrograms (mcg)
2.4
0.5
0.9
2.8
2
We updated the RDIs for the following nutrients and minerals based on their RDAs: calcium, copper, folate,
iodine, iron, magnesium, molybdenum, niacin, phosphorus, riboflavin, selenium, thiamin, vitamin A, vitamin B6,
vitamin B12, vitamin C, vitamin D, vitamin E, and zinc (81 FR 33742 at 33897 through 33901). Updated RDIs
based on AIs are available for the following nutrients and minerals: biotin, chloride, choline, chromium, manganese,
pantothenic acid, potassium, and vitamin K (81 FR 33742 at 33926).
Contains Nonbinding Recommendations
14
Nutrient
Unit of Measure
Adults
and
Children
≥ 4 years
Infants
1
through
12
months
Children
1
through
3 years
Pregnant
women
and
lactating
women
Biotin
Micrograms (mcg)
30
6
8
35
Pantothenic Acid
Milligrams (mg)
5
1.8
2
7
Phosphorus
Milligrams (mg)
1,250
275
460
1,250
Iodine
Micrograms (mcg)
150
130
90
290
Magnesium
Milligrams (mg)
420
75
80
400
Zinc
Milligrams (mg)
11
3
3
13
Selenium
Micrograms (mcg)
55
20
20
70
Copper
Milligrams (mg)
0.9
0.2
0.3
1.3
Manganese
Milligrams (mg)
2.3
0.6
1.2
2.6
Chromium
Micrograms (mcg)
35
5.5
11
45
Molybdenum
Micrograms (mcg)
45
3
17
50
Chloride
Milligrams (mg)
2,300
570
1,500
2,300
Choline
Milligrams (mg)
550
150
200
550
Protein
Grams (g)
n/a
11
n/a
71
7
1
RDIs are based on dietary reference intake recommendations for infants through 12 months of age.
2
RAE = Retinol activity equivalents; 1 microgram RAE = 1 microgram retinol, 2 micrograms supplemental β-
carotene, 12 micrograms dietary β-carotene, 24 micrograms dietary α-carotene, or 24 micrograms dietary β-
cryptoxanthin.
3
1 mg α-tocopherol (label claim) = 1 mg α-tocopherol = 1 mg RRR- α-tocopherol = 2 mg all rac- α-tocopherol.
4
NE = Niacin equivalents, 1 mg NE = 1 mg niacin = 60 milligrams tryptophan.
5
“Folate” and “Folic Acid” must be used for purposes of declaration in the labeling of conventional foods and
dietary supplements. The declaration for folate must be in mcg DFE (when expressed as a quantitative amount by
weight in a conventional food or a dietary supplement), and percent DV based on folate in mcg DFE. Folate may be
expressed as a percent DV in conventional foods. When folic acid is added or when a claim is made about the
nutrient, folic acid must be declared in parentheses, as mcg of folic acid.
6
DFE = Dietary Folate Equivalents; 1 DFE = 1 mcg naturally-occurring folate = 0.6 mcg folic acid.
7
Based on the reference caloric intake of 2,000 calories for adults and children aged 4 years and older, and for
pregnant women and lactating women.
VII.C.1 Which Units of Measure Have Changed?
The units of measure have changed for vitamin A, vitamin D, vitamin E, folate, and niacin.
Where previously “international units,” or “IU,” was used for vitamin A, vitamin D, and vitamin
E, we now require the use of a metric unit of measure (mcg RAE for vitamin A, mcg for vitamin
D, and mg α-tocopherol for vitamin E). The declarations of vitamin A and vitamin D will be
expressed in mcg on the nutrition and supplement facts labels, and vitamin E will be expressed in
mg (21 CFR 101.9(c)(8)(iv)). The amount of vitamin D may also be expressed in IU, in addition
to the mandatory declaration in mcg. Any declaration of the amount of vitamin D in IU must
appear in parentheses after its declaration in mcg (21 CFR 101.9(c)(8)(iv)). This is allowed
because vitamin D is a nutrient of public health significance, the National Academy of Medicine
(formerly the Institute of Medicine (IOM)) uses IU for vitamin D, and voluntary labeling in IU
will assist consumers in maintaining healthy dietary practices (81 FR 33742 at 33912). For
Contains Nonbinding Recommendations
15
vitamin E, the use of this new unit of measure would account for the difference in activity
between naturally occurring and synthetic vitamin E (81 FR 33742 at 33913).
Changes regarding the labeling of folate have been made so that there is now consistency
between conventional foods and dietary supplements. When the amount of folate is declared in
the labeling of a conventional food or a dietary supplement, the name “folate” is to be listed for
products containing folate (both natural and synthetic), folic acid, or a mixture of folate and folic
acid. Folate is to be declared in mcg Dietary Folate Equivalents (DFE). The terms folic acid or
folacin are no longer allowed to be used as synonyms for folate. They cannot be listed without
parentheses in place of folate, and they cannot be added in parentheses immediately following
folate, unless synthetic folic acid is added as a nutrient supplement to a product or a claim is
made about it. In that case, the declaration of folic acid must be included, in parentheses after
the declaration of folate, as a quantitative amount by weight (21 CFR 101.9(c)(8)(ii)).
The unit of measure has changed for niacin, from “mg” to “milligrams NE,” where “NE” stands
for “niacin equivalents” (21 CFR 101.9(c)(8)(iv)). However, only the amount will continue to be
declared on the Nutrition and Supplement Facts labels. The declaration of niacin content
requires products to include both preformed niacin (from nicotinic acid and nicotinamide in the
diet or niacin) and tryptophan, including those that may not contain preformed niacin.
Compliance can be determined by measuring niacin and tryptophan separately; AOAC methods
exist for both niacin and tryptophan (81 FR 33742 at 33915).
VII.C.2 How Do I Know if the Declaration of Vitamins and Minerals is Mandatory or
Voluntary?
Declaration is mandatory for the vitamins and minerals of public health significance, which have
been updated and now are vitamin D, calcium, iron, and potassium. They must be listed in that
order, and their declaration is mandatory on foods for adults and children 4 years of age and
older, as well as on foods represented or purported to be specifically for infants through 12
months, children 1 through 3 years of age, or pregnant women and lactating women (21 CFR
101.9(c)(8)(ii)).
Voluntary declaration is allowed for any other essential nutrient presented in Figure 2. However,
if any of these nutrients is added to food as a nutrient supplement or if a claim is made about it
on the label or in the labeling of foods, then mandatory declaration is required (21 CFR
101.9(c)(8)(ii)).
VII.C.3 What Information Has to be Declared for Vitamins and Minerals?
For the vitamins and minerals of public health significance (vitamin D, calcium, iron, and
potassium), the inclusion of both absolute amounts (quantitative amount by weight) and percent
DV are required (except as described for labels in 21 CFR 101.9(j)(13)). However, if these
nutrients are present at an amount less than 2 percent of the RDI, then the quantitative amounts
and percentages of these nutrients are not required to be declared. They may be declared by a
zero or by the use of an asterisk (or other symbol) that refers to another asterisk (or symbol) that
is placed at the bottom of the table and that is followed by the statement “Contains less than 2
Contains Nonbinding Recommendations
16
percent of Daily Value of this (these) nutrient (nutrients).” Alternatively, except as provided in
21 CFR 101.9(f), if vitamin D, calcium, iron, or potassium is present in amounts less than 2
percent of the RDI, label declaration of the nutrient(s) is not required if the statement “Not a
significant source of (listing the vitamins or minerals omitted)” is placed at the bottom of the
table of nutrient values (21 CFR 101.9(c)(8)(iii)).
When any other nutrient listed in Figure 2 is added to a food as a nutrient supplement or when a
claim is made about it (therefore triggering mandatory declaration), the declaration must include
the amount per serving, expressed as percent DV, unless otherwise stated as both the quantitative
amount by weight and percent DV (21 CFR 101.9(c)(8)(ii)). Any nutrient (except folic acid) that
is voluntarily declared must be declared as a percent of the RDI (21 CFR 101.9(c)(8)(ii)) but
declaring the quantitative amount by weight is voluntary (81 FR 33742 at 33947-48). The
quantitative amount of folic acid must always be declared (21 CFR 101.9(c)(8)(ii)). You must
use the appropriate RDI, depending on which population group your food is represented or
purported to be for (21 CFR 101.9(c)(8)(i)).
Zeros following the decimal points may be dropped, and additional levels of significance may be
used when the number of decimal places indicated is not sufficient to express lower amounts
(e.g., the RDI for zinc is given in whole milligrams, but the quantitative amount may be declared
in tenths of a milligram) (21 CFR 101.9(c)(8)(iii)). The phrase “levels of significance” refers to
the degree of accuracy when rounding nutrients for purposes of declaring quantitative amounts
of vitamins and minerals on the label. For example, the RDIs for some vitamins and minerals
are small numerical values, and nutrients with an RDI of less than 5 (i.e. thiamin, riboflavin,
vitamin B
6
, vitamin B
12
, copper, and manganese) would not be able to be declared on the
Supplement Facts label if they contain less than 2 percent of the RDI and the amount is declared
to the nearest mg or mcg. In such situations, manufacturers could declare the quantitative
amounts to the nearest hundredth of a mg or mcg per serving, provided that such a level of
specificity does not represent a greater level of precision in the amount of the nutrient present
than can be scientifically supported. These rounding recommendations are explained in further
detail in FDA’s guidance for industry entitled “Nutrition and Supplement Facts Labels:
Questions and Answers Related to the Compliance Date, Added Sugars, and Declaration of
Quantitative Amounts of Vitamins and Minerals” (Ref. 3).
VII.D How Do the Nutrient Requirements for Supplement Facts Labels Differ?
Both the content and format of the Supplement Facts label have been updated to correspond to
the Nutrition Facts label, wherever possible. However, there are still some differences between
the two. On the Supplement Facts label, information on calories and serving size was not made
more prominent through increased type size, as it has been on the Nutrition Facts label. While
the percentage of the RDI for protein for foods purported to be for infants through 12 months of
age is allowed on Nutrition Facts labels, this declaration must be omitted for Supplement Facts
labels (21 CFR 101.36(b)(2)(iii)).
In addition, the new ordering for vitamins and minerals on the Supplement Facts label is as
follows: vitamin A, vitamin C, vitamin D, vitamin E, vitamin K, thiamin, riboflavin, niacin,
vitamin B6, folate and folic acid, vitamin B12, biotin, pantothenic acid, choline, calcium, iron,
Contains Nonbinding Recommendations
17
phosphorus, iodine, magnesium, zinc, selenium, copper, manganese, chromium, molybdenum,
chloride, sodium, potassium, and fluoride (21 CFR 101.36(b)(2)(B)).
VII.E Has Nutrient Compliance or the Level of Variance Allowed Changed?
There are still two classes of nutrients for compliance purposes. Class I covers those added
nutrients in fortified or fabricated foods (21 CFR 101.9(g)(3)(i)), and Class II nutrients are those
that are naturally occurring, or indigenous, nutrients (21 CFR 101.9(g)(3)(ii)). For compliance
purposes, if both Class I and Class II nutrients are present in the final food product, then the total
amount of the nutrient (both indigenous and exogenous) is subject to class I requirements.
The updated rule does not change the level of variances allowed in 21 CFR 101.9(g)(4) and
101.9(g)(5), but the rule also now includes soluble fiber and insoluble fiber in 21 CFR
101.9(g)(4) and added sugars in 21 CFR 101.9(g)(5). Therefore, 21 CFR 101.9(g)(4) now states
that a food with a label declaration of a vitamin, mineral, protein, total carbohydrate, dietary
fiber, soluble fiber, insoluble fiber, polyunsaturated or monounsaturated fat will be deemed to be
misbranded under section 403(a) of the FD&C Act, unless the nutrient content of the composite
is formulated to be at least equal to the declared value for the vitamin, mineral, protein, or dietary
fiber meeting the definition of a Class I nutrient (potassium has been removed), or the nutrient
content of the composite is at least equal to 80 percent of the declared value for the vitamin,
mineral, protein, total carbohydrate, polyunsaturated or monounsaturated fat, or dietary fiber
meeting the definition of a Class II nutrient (potassium and “other carbohydrate” have been
removed). Our regulations, at 21 CFR 101.9(g)(5), state that a food with a label declaration of
calories, sugars, added sugars (when the only source of sugars in the food is added sugars), total
fat, saturated fat, trans fat, cholesterol, or sodium will be deemed to be misbranded under section
403(a) of the FD&C Act if the nutrient content of the composite is greater than 20 percent in
excess of the value for that nutrient declared on the label. Reasonable excesses of soluble fiber,
insoluble fiber and sugar alcohols, and reasonable deficiencies of added sugars are also now
acceptable within current good manufacturing practices.
VII.F Which Version of the Official Methods of Analysis of the AOAC International
Should be Used?
The final rule now incorporates by reference the Official Methods of Analysis of the AOAC
International, 19th edition (2012) (21 CFR 101.9(l)(1)(i)).
VIII. How Do I Comply with the Formatting Requirements?
There are required formats for Nutrition and Supplement Facts labels. We strongly recommend
that the nutrition information be presented using the graphic specifications below (see also
Appendix B to 21 CFR part 101 and Ref. 8).
VIII.A How Do I Comply with the Standard Version of the Nutrition Facts Label?
Figures 3 through 5 reflect what a standard vertical Nutrition Facts label must look like (see 21
CFR 101.9(d)(12)). This format is to be used except on foods where the tabular display is

Contains Nonbinding Recommendations
18
permitted, where dual columns are required or voluntarily used, where the simplified format is
used, where the aggregate display is used, and on foods in small or intermediate-sized packages.
These formats, except for the aggregate display, as no changes to this format were made, are
discussed in detail below.
Figures 3 and 4 show a Nutrition Facts label including only those nutrient declarations which are
mandatory. Figure 5 shows a Nutrition Facts label that includes both mandatory and voluntary
nutrient declarations.
Figure 3: Standard Vertical with Mandatory Nutrients
Figure 4: Standard vertical, Side-by-side
.
1. Heading set to
full width, unless
impractical
2.Thin hairline beneath
heading
3. Type size of
word “Calories”
is no smaller
than 16 point
4. Mandatory
vitamins and
minerals listed
vertically
2. Type size of
numerical value is no
smaller than 22 point
1. “Serving size”
numerical value is right
justified
3. “Added Sugars”
Declaration
4. Mandatory
vitamins and
minerals listed
side-by-side

Contains Nonbinding Recommendations
19
Figure 5: Standard Vertical, Mandatory and Voluntary Nutrients
1. Caloric
conversion
information may be
declared
VIII.A.1 How Has the Formatting Changed for the “Nutrition Facts” Header on a
Standard Vertical Label?
The “Nutrition Facts” heading must be in a type size no smaller than all other print size in the
nutrition label, except for the numerical information for “Calories,” which according to 21 CFR
101.9(d)(1)(iii) must be in a type size no smaller than 22 point for the standard label (21 CFR
101.9(d)(2)). It must be set the full width of the nutrient information, unless impractical (21
CFR 101.9(d)(2)). It is not required to be set the full width of the nutrient information for labels
presented according to the following formats: tabular display, aggregate display, tabular dual
column display, tabular display for small or intermediate-sized packages, and linear display for
small or intermediate-sized packages (21 CFR 101.9(d)(2)).
A thin horizontal line (i.e., a hairline rule) is to be inserted directly beneath the Nutrition Facts
heading, before the servings per container statement (except on the linear display discussed
below) (21 CFR 101.9(d)(1)(v)).
Contains Nonbinding Recommendations
20
VIII.A.2 How Has the Formatting Changed for the Declaration of Calories on a
Standard Vertical Label?
The type size has increased for “Calories” and the numeric value of “Calories.” On the standard
vertical display of the Nutrition Facts label, the numeric value of “Calories” must be listed in a
type size no smaller than 22 point, as well as in bold or extra bold type highlighting (21 CFR
101.9(d)(1)(iii)). The word “Calories” must be listed in a type size no smaller than 16 point, as
well as in bold or extra bold type highlighting (21 CFR 101.9(d)(1)(iii)). See Figures 3 through
5.
VIII.A.3 How Has the Formatting Changed for the Declarations of Serving Size and
Servings Per Container on a Standard Vertical Label?
The “___ servings per container” declaration now immediately follows the “Nutrition Facts”
heading and must be in a type size no smaller than 10 point (21 CFR 101.9(d)(3)(i)).
Below “___ servings per container” is the declaration of “Serving size.” “Serving size” is to be
highlighted in bold or extra bold and in a type size no smaller than 10 point (21 CFR
101.9(d)(3)(ii)). While the “Serving size” declaration is still left-justified, the corresponding
numerical value is now right-justified on Nutrition Facts labels only (not on Supplement Facts
labels), provided that adequate space is available (21 CFR 101.9(d)(3)(ii)). If the “Serving size”
declaration does not fit in the allocated space, then a type size no smaller than 8 point may be
used on packages of any size.
For further information on the updates made to the Reference Amounts Customarily Consumed
and Serving Size regulations, please reference the Small Entity Compliance Guide entitled “Food
Labeling: Serving Sizes of Foods That Can Reasonably Be Consumed At One Eating Occasion;
Dual-Column Labeling; Updating, Modifying, and Establishing Certain Reference Amounts
Customarily Consumed; Serving Size for Breath Mints; and Technical Amendments: Guidance
for Industry” (Ref. 9), as well as the final guidance entitled “Food Labeling: Serving Sizes of
Foods That Can Reasonably Be Consumed At One Eating Occasion, Reference Amounts
Customarily Consumed, Serving Size-Related Issues, Dual-Column Labeling, and Miscellaneous
Topics” (Ref. 10).
VIII.A.4 How Has the Formatting Changed for the Declaration of Vitamins and
Minerals on a Standard Vertical Label?
Nutrient information for vitamins and minerals (except sodium) are to be separated from
information regarding other nutrients by a bar (21 CFR 101.9(d)(8)). They may be listed
vertically (Figures 3 and 5) or horizontally (Figure 4). If listed horizontally in two columns, then
vitamin D and calcium should be listed on the first line, and iron and potassium should be listed
on the second line (21 CFR 101.9(d)(8)). Nutrient information must be in a type size no smaller
than 8 point (21 CFR 101.9(d)(1)(iii)).
VIII.A.5 Where Can I Declare “Calories from Saturated Fat” on a Standard Vertical
Label?
Contains Nonbinding Recommendations
21
While “Calories from fat” can no longer be declared, “Calories from saturated fat” is still
allowed (21 CFR 101.9(c)(1)(ii)). When declared, it is to be indented under the statement of
calories and must be in a type size no smaller than 8 point (21 CFR 101.9(d)(5)).
VIII.A.6 Where, and How, Do I Declare “Added Sugars” on a Standard Vertical
Label?
The statement “Includes ‘X’ g Added Sugars” must be used to declare added sugars content
(except on packages of single ingredient sugars), and this statement must be indented directly
beneath the listing for “Total Sugars” on the Nutrition Facts label (21 CFR 101.9(c)(6)(iii)). The
‘X’ will be filled in with the respective added sugar content (21 CFR 101.9(c)(6)(iii)). In
Appendix B to 21 CFR 101, we illustrate our recommendation to shorten the length of the
hairline requirement between total sugars and added sugars to help denote that “added sugars”
are a subcomponent of “total sugars.” As mentioned above, a DRV has been established for
added sugars for most population groups, and the % Daily Value declaration is required as well
(21 CFR 101.9(d)(7)(ii)). Added sugars must also be declared on the Supplement Facts label (21
CFR 101.36(b)(2)(i)).
Added sugars content must be expressed to the nearest gram, except in the following
circumstances:
• The statement “Contains less than 1 gram” or, alternatively, “less than 1 gram” may be
used if a serving contains less than 1 gram added sugars; or
• The added sugars content may be expressed as zero if the serving contains less than 0.5
gram.
See 21 CFR 101.9(c)(6)(iii).
If a product contains less than 1 gram of added sugars per serving and no claims are made about
sweeteners, sugars, or sugar alcohol content, then the declaration of the amount of added sugars
is not required. If the added sugars content is therefore not declared, you must place the
statement “Not a significant source of added sugars” at the bottom of the table of nutrient values
(except as provided for in 101.9(f)) (21 CFR 101.9(c)(6)(iii)).
VIII.A.7 How Has the “% Daily Value*” Footnote Been Changed?
The final rule modified the footnote that refers to the “% Daily Value*” column heading to
provide clarity and a better explanation. The modified footnote now states that “*The % Daily
Value tells you how much a nutrient in a serving of food contributes to a daily diet. 2,000
calories a day is used for general nutrition advice” on all Nutrition Facts label formats on foods
for the general population (21 CFR 101.9(d)(9)), with the exception of foods having minimal
calories and with certain formats (i.e., the simplified display, infants through 12 months of age,
children 1-3 years, and the linear and tabular displays for small or intermediate-sized packages),
as described below. The footnote table listing DRVs for total fat, saturated fat, cholesterol,
Contains Nonbinding Recommendations
22
sodium, total carbohydrate, and dietary fiber for 2,000 and 2,500 calorie diets is no longer
included as part of this footnote (81 FR 33742 at 33950).
The footnote may be omitted for products that can use the terms “calorie free,” “free of calories,”
“without calories,” “trivial source of calories,” “negligible source of calories,” or “dietary
insignificant source of calories” on the label or in the labeling of foods. However, the voluntary
use of the first part of the footnote (“*The % Daily Value tells you how much a nutrient in a
serving of food contributes to a daily diet.”) is allowed for these foods (21 CFR 101.9(d)(9)).
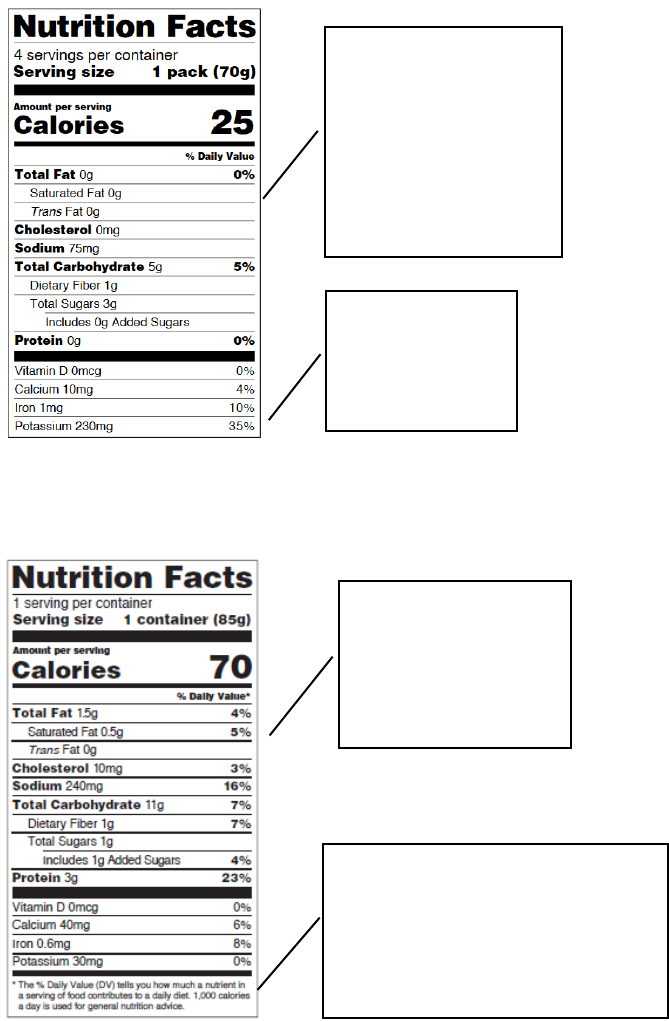
VIII.A.8 What Changes Need to be Made on Labels for Foods Represented or
Purported to be Specifically for Infants Through 12 Months of Age or for
Children 1 Through 3 Years of Age?
The next two figures reflect the Nutrition Facts labels for foods represented or purported to be
specifically for infants through 12 months of age (Figure 6) and for children 1 through 3 years of
age (Figure 7). The nutrients declared for these subgroups must include calories, total fat,
saturated fat, trans fat, cholesterol, sodium, total carbohydrate, dietary fiber, total sugars, added
sugars, protein, vitamin D, calcium, iron, and potassium (21 CFR 101.9(j)(5)(i)). Calories from
saturated fat, polyunsaturated fat, and monounsaturated fat can now be voluntarily declared, if
desired (see 21 CFR 101.9(c)).
Percent DVs for total fat and potassium on the labeling of foods, other than infant formula,
represented or purported to be for infants through 12 months of age are now required (see 21
CFR 101.9(d)(7) and (j)(5)). However, a percent DV declaration for saturated fat, cholesterol,
sodium, dietary fiber, and added sugars is not allowed on such foods. In addition, as with foods
for all other populations, a percent DV declaration for trans fat and total sugars is not allowed, as
no DRVs have been established for those nutrients (21 CFR 101.9(j)(5)(ii)(A)). No “% Daily
Value*” footnote is to be declared either (21 CFR 101.9(j)(5)(ii)(A)).
On labels of foods, other than infant formula, represented or purported to be for children 1
through 3 years of age, the “% Daily Value*” footnote statement must read: “*The % Daily
Value tells you how much a nutrient in a serving of food contributes to a daily diet. 1,000
calories a day is used for general nutrition advice” (21 CFR 101.9(d)(9)), and a type size no
smaller than 6 point must be used for the footnote (21 CFR 101.9(d)(1)(iii)).
Contains Nonbinding Recommendations
23
Figure 6: Infants through 12 Months of Age (21 CFR 101.9(j)(5)(ii)(B))
1. No % Daily Value is
declared for: saturated
fat, trans fat, cholesterol,
sodium, dietary fiber,
total sugars, or added
sugars
2. N
ote the absence
of a footnote at the
bottom
Figure 7: Children 1 through 3 Years (21 CFR 101.9(j)(5)(ii)(B))
1. % Daily Value
declared for the required
nutrients per 21 CFR
101.9(c)(9)
2. The footnote replaces 2,000 calories
with 1,000 calories
And
3. Type size for footnote no smaller
than 6 point
VIII.B When Can I Use the Tabular Format?
The tabular format can be used when there is not sufficient continuous vertical space to
accommodate the required components of the nutrition label, up to and including the mandatory
declaration of potassium (21 CFR 101.9(d)(11)(iii)). This is different than the tabular format for

Contains Nonbinding Recommendations
24
small or intermediate-sized packages (see Section VIII.C below). Figure 8 reflects the tabular
format.
Figure 8: Tabular Display
1. “Nutrition Facts” header is
not set across the entire width
of the label
2. Quantitative amounts of Thiamin,
Riboflavin, and Niacin are not declared,
as only % DV is required if voluntarily
declared
VIII.B.1 How Does the Declaration of Calories in the Tabular Format Differ from
that of the Standard Vertical Label Version?
In the tabular format, the word “Calories” must be in a type size no smaller than 10 point, instead
of 16 point as required for the standard vertical label (21 CFR 101.9(d)(1)(iii)). The 10-point
type size for calories is also required for the tabular display for small and intermediate-sized
packages (see section VIII.C) and the tabular dual column display (see section VIII.D) (21 CFR
101.9(d)(1)(iii)).
VIII.B.2 How Do the Declarations of Serving Size and Servings Per Container in the
Tabular Format Differ from the Standard Vertical Label Version?
The “___ servings per container” declaration is the same as required for the standard vertical
label version. However, the declaration of “Serving size,” which is listed below the “___
servings per container” declaration, must be highlighted in bold or extra bold and in a type size
no smaller than 9 point (21 CFR 101.9(d)(3)(i)). This format is also the same for the tabular
display for small and intermediate-sized packages (see section VIII.C) and the tabular dual
column display (see section VIII.D) (21 CFR 101.9(d)(3)(i)).
VIII.C How Do I Comply with These Requirements if My Food Packaging Only Has a
Total Surface Area Available to Bear Labeling of 40 or Less Square Inches?
We allow modifications of the Nutrition Facts label for foods in packages with a total surface
area available to bear labeling of 40 or less square inches. One modification is allowing the use
of the tabular display for small packages or intermediate-sized packages if the product has a total
surface area available to bear labeling of 40 or less square inches and neither the standard
vertical display (see Figures 3-5) nor the tabular display (see Figure 8) can be accommodated (21
CFR 101.9(j)(13)(ii)(A)). See Figure 9.

Contains Nonbinding Recommendations
25
Figure 9: Tabular Display for Small Packages
Another modification is the use of a linear display. For products with a total surface area
available to bear labeling of 40 or less square inches, nutrition information can be presented in a
linear format, but only if the label will not accommodate a tabular display (21 CFR
101.9(j)(13)(ii)(A)). However, when there is less than 12 square inches of available labeling
space, either the small tabular or linear display can be used (21 CFR 101.9(j)(13)(ii)(A)). We
retained the preexisting linear label format to provide flexibility for labels on small packages
with various shapes and sizes. However, we adapted it to maintain consistency with the other
formatting changes that have been finalized. As discussed in detail below, the “Calories”
information has increased in type size (see Section VIII.C.1) and the serving size and serving per
container declarations have been updated (see Section VIII.C.2). In addition, “Sugars” has
changed to “Total Sugars” (21 CFR 101.9(d)(6)(ii)), “Added Sugars” declaration is now
mandatory (21 CFR 101.9(d)(6)(iii)), and the abbreviated footnote “% DV = % Daily Value” is
optional on some formats (see Section VIII.C.3). Note that the hairline that separates the
“Nutrition Facts” header from the rest of the label is not present on the linear display. Figure 10
shows the updated linear format.
Figure 10: Linear Display for Small and Intermediate-Sized Packages
(101.9(j)(13)(ii)(A)(2))
1. “Nutrition Facts” header is
not set across the entire width
of the label
2. “Serv. size,” “Sat. fat,” “Cholest.,” “Total
carb.,” “Fiber,” “Incl.,” “Vit.,” and “Potas.”
are all acceptable abbreviations for the
linear display
3. The abbreviated footnote “% DV = %
D
aily Value” is not shown on this label,
as it is not mandatory (see Section
VIII.D.3)
Contains Nonbinding Recommendations
26
The third modification is the use of abbreviations, as listed in 21 CFR 101.9(j)(13)(ii)(B). We
now allow for the following additional abbreviations on labels for small and intermediate-sized
packages: “vitamin” may be abbreviated as “Vit.,” “potassium” may be abbreviated as “Potas.,”
and “Includes” may be abbreviated as “Incl.” These abbreviations will further conserve label
space. In addition, we allow the abbreviations of “Total carb.” and “Incl.” to be used on dual-
column display labels (see section VIII.D).
VIII.C.1 What Type Size Must be Used for the Declaration of Calories on the Tabular
or Linear Displays for Small or Intermediate-Sized Packages?
The numeric value for “Calories” must be in a type size no smaller than 14 point, and the word
“Calories” must be in a type size no smaller than 10 point (21 CFR 101.9(d)(1)(iii)). See Figures
9 and 10.
VIII.C.2 How Do the Declarations of Serving Size and Servings Per Container on the
Tabular or Linear Displays for Small or Intermediate-Sized Packages Differ
from the Standard Label Version?
The “___ servings per container” declaration immediately follows the “Nutrition Facts” heading,
as in the standard label version; however, it must be in a type size no smaller than 9 point on the
tabular or linear display for small packages (21 CFR 101.9(d)(3)(i)). If a linear display is used,
then the actual number of servings (i.e., “Servings” as shown in Fig 10) may be listed instead of
the servings per container declaration.
The declaration of “Serving size,” which follows below the “___ servings per container” or
“servings” declaration, is to be highlighted in bold or extra bold and in a type size no smaller
than 9 point on the tabular or linear display for small or intermediate-sized packages. See
Figures 9 and 10.
VIII.C.3 Is the “% Daily Value*” Footnote Required on Foods in Small and
Intermediate-Sized Packages?
For products in small and intermediate-sized packages that qualify for the use of the tabular or
linear format as specified in 21 CFR 101.9(j)(13)(ii)(A)(1) and (2), there is no longer a
requirement to place an asterisk, followed by the statement “Percent Daily Values are based on a
2,000 calorie diet,” at the bottom of the label if the footnote corresponding to “% Daily Value” is
omitted. If “Daily Value” is not spelled out in the heading, then an asterisk can be placed at the
bottom of the label followed by the statement “% DV = % Daily Value” in a type size no smaller
than 6 point (21 CFR 101.9(d)(1)(iii)). See Figures 9 and 10.
VIII.D How Do I Comply with the Nutrition Labeling Formatting Requirements when Dual
Column Labeling is Required or Provided Voluntarily?
Dual column labeling is required under certain conditions, and can be used in other situations. In
the Federal Register of March 2, 2018 (83 FR 9003), we announced the availability of a separate
Small Entity Compliance Guide entitled “Food Labeling: Serving Sizes of Foods That Can
Contains Nonbinding Recommendations
27
Reasonably Be Consumed At One Eating Occasion; Dual-Column Labeling; Updating,
Modifying, and Establishing Certain Reference Amounts Customarily Consumed; Serving Size
for Breath Mints; and Technical Amendments: Guidance for Industry,” which, among other
topics, discusses a new requirement regarding the use of dual-column labeling (Ref. 9). In the
Federal Register of December 31, 2019 (84 FR 72230), we announced the availability of a final
guidance for industry entitled “Food Labeling: Serving Sizes of Foods That Can Reasonably Be
Consumed At One Eating Occasion, Reference Amounts Customarily Consumed, Serving Size-
Related Issues, Dual-Column Labeling, and Miscellaneous Topics.” This final guidance also
discusses formatting issues for dual-column labeling (Ref. 10).
Regarding specific formatting for dual column labeling, the final rule updates certain
requirements. Directly following the serving size declarations, each column of nutrition
information must contain a heading that accurately describes why a dual column is being
utilized. For example, a product may provide nutrition information for two different RDI
groups, like in Figure 11 below. A product may also provide nutrition information for two
different forms of the same food (e.g., “Per ¼ cup mix” and “Per prepared portion”), as in Figure
12 below (21 CFR 101.9(e)). The quantitative information by weight and percent DV must be
presented for each column, and the columns are to be separated by vertical lines (21 CFR
101.9(e)(3)). Nutrient information for vitamins and minerals must be separated from the
information on other nutrients by a bar and be arrayed vertically in the required order (21 CFR
101.9(e)(4)). Note that the “Amount per serving” statement is not required for the dual column
formats (21 CFR 101.9(d)(4)). See Figures 11 through 15 (see also 21 CFR 101.9(e)(5) and
(e)(6)(i)).

Contains Nonbinding Recommendations
28
Figure 11: Dual Column Display for 2 different RDI Groups (101.9(e)(5))
1. Serving Size and
Servings Per
Container reflect the
different RDI
groups for the
product
2. Abbreviations of
“Total Carb.” and
“Incl.” are allowed
on this label
3. Vitamins and
minerals are
separated from
information on other
nutrients by a bar
4. The appropriate
“% Daily Value”
footnote for each
population group is
included

Contains Nonbinding Recommendations
29
Figure 12: Dual Columns, Two Forms of the Same Food (101.9(e)(5))
1. “Amount per
serving” declaration
is not required
Figure 13: Dual Column Display, Per Serving and Per Container (101.9(e)(6)(i))
1. Two column
headings describing
why the dual
column display is
being used

Contains Nonbinding Recommendations
30
Figure 14: Dual Column Display, Per Serving and Per Unit (101.9(e)(6)(i))
Figure 15 shows the use of dual columns in a tabular display format for products. As mentioned
above, “Calories” must be in a type size no smaller than 10 point, and the numeric amount for
“Calories” must be in a type size no smaller than 22 point (21 CFR 101.9(d)(1)(iii)). “Serving
size” declaration must be in a type size no smaller than 9 point; however, the “___ servings per
container” declaration must be in a type size no smaller than 10 point, as with the standard label
version (21 CFR 101.9(d)(3)(i) and (ii)). “Amount per serving” is not required (21 CFR
101.9(d)(4)).
Figure 15: Tabular Dual Column Display (101.9(e)(6)(ii))
VIII.E When Can the Simplified Format Be Used?
You can use the simplified format when a food product contains insignificant amounts of eight
or more of the following: Calories, total fat, saturated fat, trans fat, cholesterol, sodium, total
carbohydrate, dietary fiber, total sugars, added sugars, protein, vitamin D, calcium, iron, and
potassium (21 CFR 101.9(f)). For foods intended for infants through 12 months of age and
children 1 through 3 years of age, you can use the simplified format when a food product
contains insignificant amounts of six or more of the following: Calories, total fat, sodium, total

Contains Nonbinding Recommendations
31
carbohydrate, dietary fiber, total sugars, added sugars, protein, vitamin D, calcium, iron, or
potassium (21 CFR 101.9(f)). The definition of “insignificant amount” has not changed.
The footnote requirement which states, “Not a significant source of ___” (listing the name(s) of
any nutrients listed in this paragraph that are present in insignificant amounts) still exists (21
CFR 101.9(f)(4)). While the full “% Daily Value” footnote (see 21 CFR 101.9(d)(9)) is not
required for the simplified format, the column header “% Daily Value” can be abbreviated as “%
DV.” If this abbreviation is used, then an asterisk must be placed at the end of the abbreviation
and the corresponding footnote must read, “% DV = % Daily Value” (21 CFR 101.9(f)(5)). No
footnote is required if “Daily Value” is spelled out in the column header (21 CFR 101.9(f)(5)).
Figure 16 displays an example of a simplified format label.
Figure 16: Simplified Format Label (101.9(f))
VIII.F Are Foods in Small Packages with a Total Surface Area Available to Bear Labeling
of Less than 12 Square Inches Exempt from the Nutrition Facts Labeling
Requirements?
Not always. Foods in small packages that have a total surface area available to bear labeling of
less than 12 square inches are not exempt from bearing a Nutrition Facts label if a nutrition claim
or other nutrition information in any context on the label or in labeling or advertising (21 CFR
101.9(j)(13)(i)) is used or as outlined for covered vending machine food (see 21 CFR 101.8(c)).
However, as previously allowed, if your product qualifies for and uses the exemption (i.e.,
claims or other nutrition information are not presented on the label or in labeling or advertising),
then you must list an address or telephone number that a consumer can use to obtain the required
nutrition information (21 CFR 101.9(j)(13)(i)(A)).
VIII.G How Do These New Formatting Rules Affect the Percentage Juice Declaration?
Contains Nonbinding Recommendations
32
With the changes to some type size requirements on the Nutrition Facts label, it is permissible if
the percentage juice declaration for foods purporting to be beverages that contain fruit or
vegetable juice is smaller in height than the declaration of “Serving size,” “Calories,” and the
numerical value for “Calories,” in addition to already being permitted to be smaller in height
than the brand name, product name, logo, universal product code, and “Nutrition Facts” heading
(21 CFR 101.30(e)(2)).
VIII.H Are There Specific Formatting Requirements for Supplement Facts Labels?
Information other than the title, headings, and footnotes must be in uniform type size no smaller
than 8 point (21 CFR 101.36(e)). Column headings (e.g., “Amount Per Serving” and “% Daily
Value”) and footnotes (e.g., “Percent Daily Values are based on a 2,000 calorie diet”) must be in
a type size no smaller than 6 point (21 CFR 101.36(e)). Because many dietary supplement
products may contribute a negligible amount of calories, we do not require information about
calories to be displayed in a larger type size or highlighted on Supplement Facts labels (81 FR
33742 at 33939).
For small packages which have a total surface area available to bear labeling of less than 12
square inches, all information within the nutrition label must be in type size no smaller than 4.5
point (21 CFR 101.36(i)(2)(i)).
Intermediate-sized packages, which have from 12 to 40 square inches of surface area available to
bear labeling, have different type size requirements. All information within the nutrition label
must be in a type size no smaller than 6 point (21 CFR 101.36(i)(2)(ii)). However, type size no
smaller than 4.5 point may be used on packages that have less than 20 square inches available for
labeling and more than eight dietary ingredients to be listed (21 CFR 101.36(i)(2)(ii)). Similarly,
type size no smaller than 4.5 point may be used on packages that have 20 to 40 square inches
available for labeling and more than 16 dietary ingredients to be listed (21 CFR 101.36(i)(2)(ii)).
The footnote stating that the “Percent Daily Values are based on a 2,000 calorie diet” is required
if the percent DV is declared for total fat, saturated fat, total carbohydrate, dietary fiber, protein,
or added sugars (21 CFR 101.36(b)(2)(iii)(D)). On labels of products represented or purported to
be for use by children 1 through 3 years of age, the footnote statement must read: “Percent Daily
Values are based on a 1,000 calorie diet” if the % Daily Value is declared for total fat, total
carbohydrate, dietary fiber, protein, or added sugars (21 CFR 101.36(b)(2)(iii)(D)).
If there is inadequate space to list the required information vertically, the list may be split. The
list to the right must be set off by a line that distinguishes it and sets it apart from the dietary
ingredients and percent DV information given to the left (21 CFR 101.36(e)(12)). The column
headings are also repeated (21 CFR 101.36(e)(12)). Figure 17 reflects this label formatting.

Contains Nonbinding Recommendations
33
Figure 17: Supplement Facts Label – Split List (101.36(e)(12))
IX. When Must I Comply with the Rule?
If you have $10 million or more in annual food sales, your compliance date is January 1, 2020.
If you have less than $10 million in annual food sales, your compliance date is January 1, 2021.
If you manufacture single-ingredient packages and/or containers of pure honey, pure maple
syrup, or other pure sugars and syrups, as well as the cranberry products discussed in the Final
Guidance (Ref. 1), we intend to exercise enforcement discretion until July 1, 2021, for
compliance with the labeling changes outlined in the Nutrition Facts label rule and the Serving
Size rule.
If you manufacture certain dried cranberry products with added flavorings (Ref. 11), we intend to
exercise enforcement discretion until July 1, 2020, for compliance with the labeling changes
outlined in the Nutrition Facts label final rule and the Serving Size final rule.
X. Why Must I Comply with the Rule?
Failure to comply with the final rule will render the covered food misbranded under section
403(q) of the FD&C Act and potentially other sections as well. The introduction or delivery for
introduction into interstate commerce of any food that is misbranded constitutes a prohibited act
under section 301(a) of the FD&C Act. Among potential consequences, committing a prohibited
act can result in injunction and/or seizure (see sections 302 and 304 of the FD&C Act (21 U.S.C.
332 and 334)).

Contains Nonbinding Recommendations
34
XI. References
The following references are on display at the Dockets Management Staff ((HFA-305), Food and
Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852) and are available for
viewing by interested persons between 9 a.m. and 4 p.m., Monday through Friday; they are also
available electronically at https://www.regulations.gov. FDA has verified the website addresses,
as of the date this document publishes in the Federal Register, but websites are subject to change
over time.
1. U.S. Food and Drug Administration. 2019. The Declaration of Added Sugars on Honey,
Maple Syrup, Other Single-Ingredient Sugars and Syrups, and Certain Cranberry Products:
Guidance for Industry. Accessed online at https://www.fda.gov/media/127928/download.
2. U.S. Food and Drug Administration. 2019. The Declaration of Allulose and Calories from
Allulose on Nutrition and Supplement Facts Labels: Guidance for Industry. Accessed online
at https://www.fda.gov/media/123342/download.
3. U.S. Food and Drug Administration. 2019. Nutrition and Supplement Facts Labels:
Questions and Answers Related to the Compliance Date, Added Sugars, and Declaration of
Quantitative Amounts of Vitamins and Minerals: Guidance for Industry. Accessed online at
https://www.fda.gov/media/117402/download.
4. U.S. Food and Drug Administration. 2018. The Declaration of Certain Isolated or Synthetic
Non-Digestible Carbohydrates as Dietary Fiber on Nutrition and Supplement Facts Labels:
Guidance for Industry. Accessed online at https://www.fda.gov/media/113663/download.
5. U.S. Food and Drug Administration. 2019. “FDA Grants Citizen Petition for Dietary Fiber.”
Accessed online at https://www.fda.gov/food/cfsan-constituent-updates/fda-grants-citizen-
petition-dietary-fiber.
6. U.S. Food and Drug Administration. 2020. “FDA Grants Citizen Petition on Glucomannan as
a Dietary Fiber.” Accessed online at https://www.fda.gov/food/cfsan-constituent-updates/fda-
grants-citizen-petition-glucomannan-dietary-fiber.
7. U.S. Food and Drug Administration. 2018. Scientific Evaluation of the Evidence on the
Beneficial Physiological Effects of Isolated or Synthetic Non-Digestible Carbohydrates
Submitted as a Citizen Petition (21 CFR 10.30): Guidance for Industry. Accessed online at
https://www.fda.gov/media/101183/download.
8. U.S. Food and Drug Administration. 2019. The New Nutrition Facts Label: Examples of
Different Label Formats. Accessed online at https://www.fda.gov/media/99151/download.
9. U.S. Food and Drug Administration. 2018. Food Labeling: Serving Sizes of Foods That Can
Reasonably Be Consumed At One Eating Occasion; Dual-Column Labeling; Updating,
Modifying, and Establishing Certain Reference Amounts Customarily Consumed; Service

Contains Nonbinding Recommendations
35
Size for Breath Mints; and Technical Amendments: Guidance for Industry. Accessed online
at https://www.fda.gov/media/111144/download.
10. U.S. Food and Drug Administration. 2019. Food Labeling: Serving Sizes of Foods That Can
Reasonably Be Consumed At One Eating Occasion, Reference Amounts Customarily
Consumed, Serving Size-Related Issues, Dual-Column Labeling, and Miscellaneous Topics:
Guidance for Industry. Accessed online at https://www.fda.gov/media/133699/download.
11. U.S. Food and Drug Administration. 2019. Policy Related to Cranberry Products with Added
Flavorings: Guidance for Industry. Accessed online at
https://www.fda.gov/media/130373/download.
